|
Feel free to add a custom message for people screened out by clicking Customize.This determines that if the branch conditions are met (respondent doesn’t consent), they will be sent to the end of the survey instead of being asked to complete it. Consent Form Templates Consent Addendum Template Database/Repository Consent Template DUHS Sample Consent DUHS Sample Consent (Spanish) MO345 Template. This form is for people who wish to authorize the Access to Information and Privacy (ATIP) Division at Immigration, Refugees and Citizenship Canada (IRCC) to release their personal information to an individual that they designate. Who should use this form Where children under 18 years of age intend to travel to Australia, their non-accompanying parent(s) or person(s) with parental. Click the Add a New Element Here that is indented under the branch. The consent form was updated in February 2018. At BMJ we require a signed BMJ consent form for any manuscript (or other content) that includes identifiable information about an individual. 
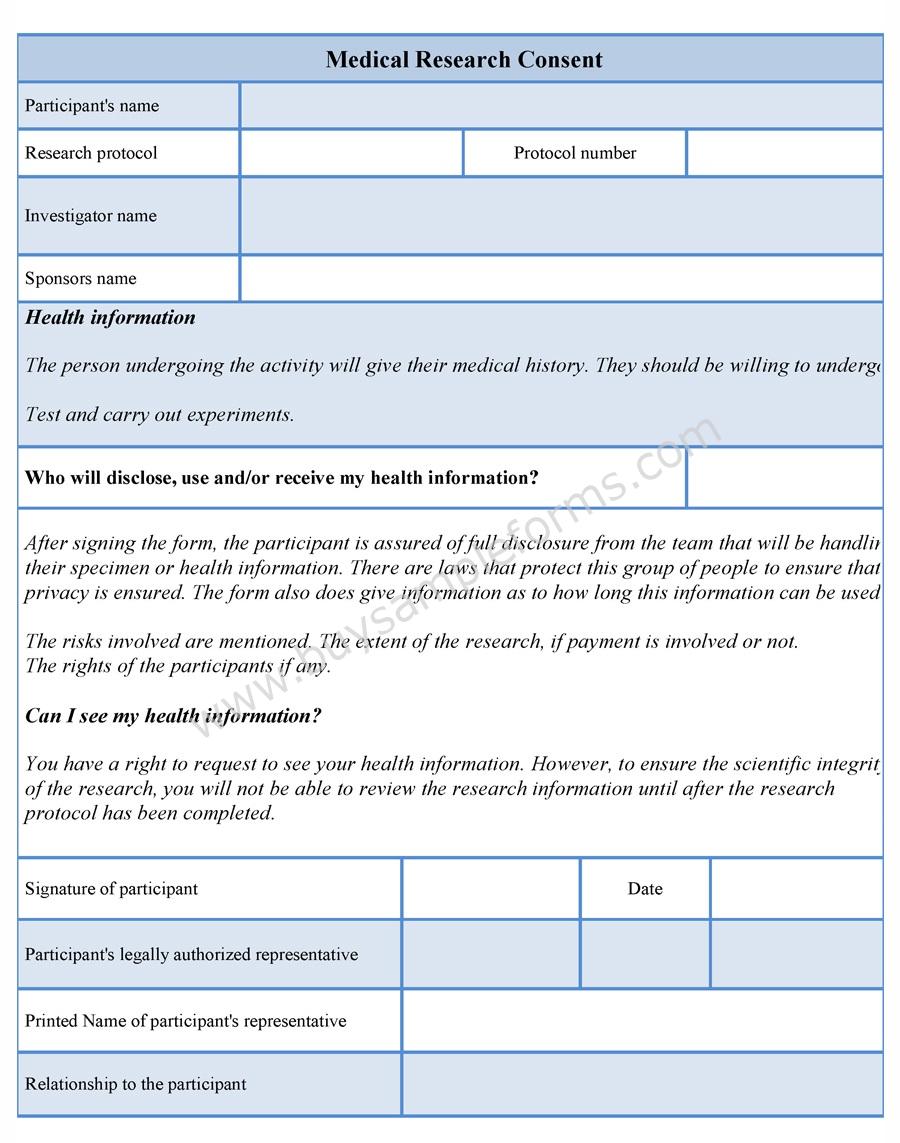
It contains a statement or declaration confirming that the patient is fully aware of any side effects and risks involved, and agrees to participate in the treatment plan. Include the project title on all pages of the consent form. Signed/Written Consent Signed consent is a written agreement between the patient and the medical practitioner.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
